Your FDA Subject Matter Expert — On Demand
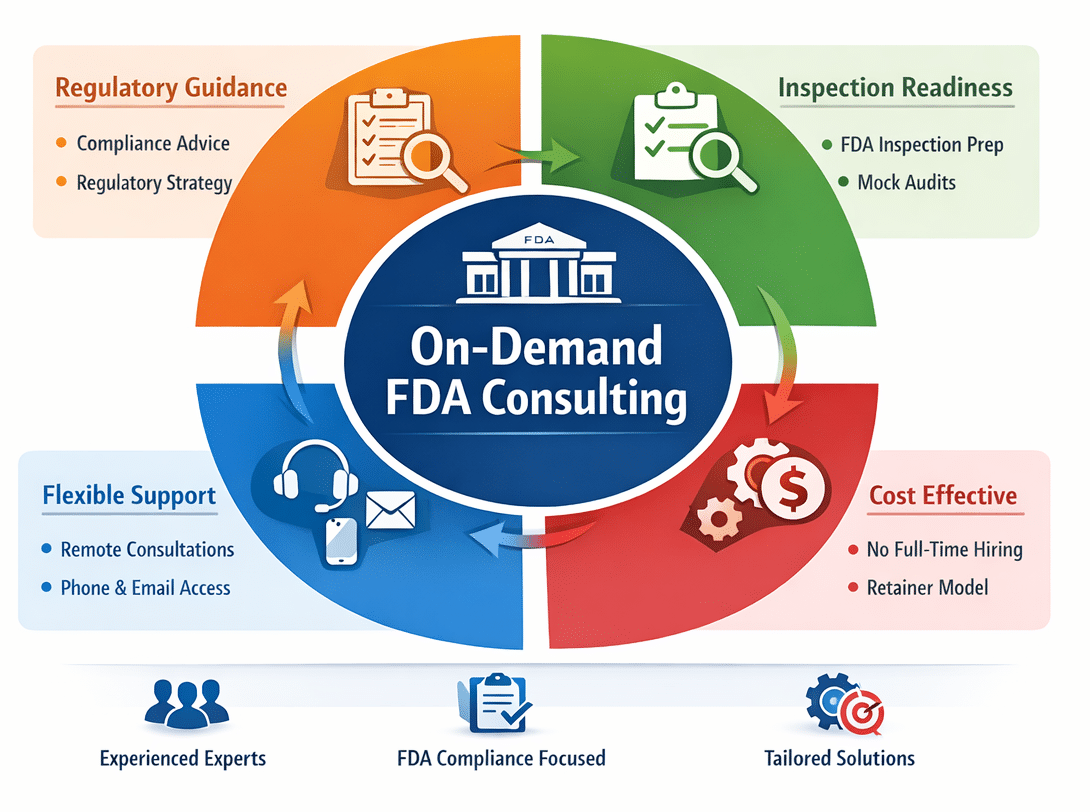
On-Demand FDA Consulting
Manufacturers frequently encounter complex regulatory questions, but do not require a full-time former FDA investigator or senior regulatory executive. Hiring experienced regulatory professionals or former FDA personnel can cost $350,000–$500,000 annually, even though many organizations need expert guidance only periodically, when critical regulatory decisions arise.
CIRG provides FDA consulting support designed to give manufacturers ongoing access to experienced regulatory insight without the cost of full-time hiring. Our team assists organizations with FDA inspection readiness, regulatory interpretation, compliance questions, and strategic regulatory planning.
Support is available when your team needs it, through remote consultations, email communication, phone discussions, and scheduled strategy meetings. This flexible approach allows manufacturers to access reliable FDA regulatory guidance while maintaining efficient operational costs.
Insighful
FDA Expertise Without the Full-Time Cost
Access to experienced FDA Compliance and regulatory expertise can significantly strengthen a manufacturer’s compliance strategy, but maintaining a full-time regulatory executive is not always necessary. Many organizations only require expert guidance when important regulatory decisions arise, during preparation for FDA inspections, or when addressing complex compliance questions. CIRG’s on-demand consulting model provides manufacturers with access to knowledgeable FDA insight without the financial burden of full-time hiring. By offering flexible consulting support, organizations can obtain reliable regulatory guidance when it is needed while maintaining efficient operational and personnel costs.
Resources
Are You Giving Enough Resources to Your Quality and Regulatory Team?
Quality and regulatory teams are responsible for navigating complex FDA requirements while maintaining daily operational responsibilities. However, many organizations expect internal teams to manage regulatory strategy, inspection readiness, and compliance questions without adequate external support. When critical regulatory decisions arise, access to experienced FDA insight can make a significant difference. Without the right resources, teams may face uncertainty when interpreting FDA expectations, preparing for inspections, or addressing compliance challenges. Providing your quality and regulatory teams with reliable regulatory guidance helps strengthen internal decision-making, improve inspection readiness, and ensure that important regulatory questions are addressed with confidence.
Compliance with Confidence
Where Does Your Team Turn When FDA Questions Arise?
When FDA-related questions arise, many quality and regulatory teams must quickly interpret regulations, guidance documents, and inspection expectations while managing their daily responsibilities. Without access to experienced regulatory insight, teams may spend valuable time trying to determine how the FDA may view a specific situation. Questions related to inspection readiness, regulatory interpretation, quality system compliance, or submission strategy can significantly impact operational decisions. Having reliable access to experienced FDA regulatory expertise provides organizations with the ability to evaluate issues more confidently, reduce uncertainty, and make informed compliance decisions when important regulatory questions arise.
The Cost of Hiring Former FDA Experts Full-Time
Hiring senior regulatory professionals or former FDA personnel can be a significant investment for many manufacturers. Experienced regulatory executives and former FDA experts often command salaries ranging from $350,000 to $500,000 annually when considering compensation, benefits, and long-term employment commitments. However, many organizations do not require this level of expertise on a full-time basis. In many cases, regulatory insight is needed periodically when complex compliance questions arise, when preparing for FDA inspections, or when developing a regulatory strategy. Maintaining access to high-level regulatory expertise without the financial burden of full-time hiring allows manufacturers to manage regulatory risks while maintaining efficient operational costs.
A Practical Alternative: On-Demand FDA Consulting
On-demand FDA consulting provides manufacturers with access to experienced regulatory insight without the cost of maintaining a full-time regulatory executive. CIRG offers a flexible consulting retainer that allows organizations to obtain expert guidance when regulatory questions arise or when strategic decisions require an experienced FDA perspective. Our team supports manufacturers with regulatory interpretation, inspection readiness, compliance questions, and mock FDA inspection preparation. This model allows companies to access reliable FDA consulting support through scheduled consultations, email communication, phone discussions, and remote meetings. By providing regulatory expertise only when needed, organizations can strengthen compliance programs while maintaining efficient operational resources.
Benefits of On-Demand FDA Consulting
On-demand FDA consulting provides manufacturers with flexible access to experienced regulatory expertise without the cost and commitment of full-time hiring. Organizations can obtain guidance when critical regulatory questions arise, during preparation for FDA inspections, or when evaluating compliance and regulatory strategy decisions. This approach allows quality and regulatory teams to strengthen internal capabilities while receiving support from professionals familiar with FDA expectations and inspection practices. By accessing expert insight when needed, companies can respond to regulatory challenges more confidently, improve inspection readiness, and maintain consistent alignment with evolving FDA regulatory requirements while managing operational resources efficiently.
How CIRG’s Consulting Retainer Works
CIRG’s consulting retainer provides manufacturers with ongoing access to experienced FDA regulatory insight without the need for full-time employment. Under this model, organizations can reach out whenever regulatory questions arise or when additional expertise is needed to support compliance decisions. Our team provides guidance on FDA inspection readiness, regulatory interpretation, quality system compliance, and strategic regulatory planning. Support is available through remote consultations, email communication, phone discussions, and scheduled strategy meetings. This flexible structure allows companies to obtain timely regulatory input while maintaining control over operational costs and internal resource allocation.
Retainer fees are determined based on several factors, including the risk classification of the product, the size and complexity of manufacturing operations, the complexity of the manufacturing process, and the current state of regulatory compliance. Organizations interested in this consulting model are encouraged to contact CIRG to discuss their specific needs and determine an appropriate retainer structure.
Support When You Need It
Regulatory challenges and compliance questions do not always arise on a predictable schedule. Manufacturers often need timely regulatory guidance when preparing for inspections, interpreting FDA expectations, or addressing complex compliance issues. CIRG’s on-demand consulting model allows organizations to access experienced regulatory support whenever it is needed. Our team is available through remote consultations, email communication, phone discussions, and scheduled virtual meetings to provide practical regulatory insight. Whether your team requires assistance with inspection readiness, regulatory interpretation, or strategic compliance decisions, CIRG provides reliable support to help organizations respond confidently to evolving regulatory requirements.
Why You Choose CIRG
Manufacturers choose CIRG because we combine practical regulatory insight with experience in how FDA evaluates compliance, inspections, and quality systems. Our consulting approach focuses on providing clear, actionable guidance that helps organizations address regulatory questions, strengthen compliance programs, and improve inspection readiness. With flexible on-demand consulting support, companies can access experienced regulatory expertise when it matters most without the cost of maintaining a full-time regulatory executive. By working alongside quality and regulatory teams, CIRG helps manufacturers navigate complex regulatory challenges with greater confidence and clarity.

